Advertisements
Advertisements
प्रश्न
Match the reactions given in Column I with the names given in Column II.
| Column I | Column II | |
| (i) | 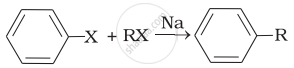 |
(a) Fittig reaction |
| (ii) | 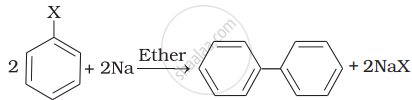 |
(b) Wurtz Fittig reaction |
| (iii) | 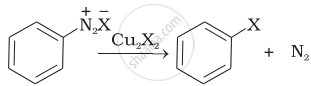 |
(c) Finkelstein reaction |
| (iv) | \[\ce{C2H5Cl + Nal ->[dry acetone] C2H5l + NaCl}\] | (d) Sandmeyer reaction |
उत्तर
| Column I | Column II | |
| (i) | 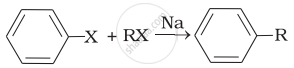 |
(b) Wurtz Fittig reaction |
| (ii) |  |
(a) Fittig reaction |
| (iii) |  |
(d) Sandmeyer reaction |
| (iv) | \[\ce{C2H5Cl + Nal ->[dry acetone] C2H5l + NaCl}\] | (c) Finkelstein reaction |
Explanation:
(i) A mixture of an alkyl halide and aryl halides gives an alkylarene when treated with sodium in dry ether and this is called Wurtz-Fittig reaction.
(ii) Aryl halides give analogous compounds when treated with sodium in dry ether, in which two aryl groups are joined together. It is called Fitting reaction.
(iii) Diazonium salt when treated with cuprous chloride or cuprous bromide gives chlorobenzene or bromobenzene. The reaction is known as Sandmeyer’s reaction.
(iv) Alkyl iodides are prepared by the reaction of alkyl chlorides with sodium iodide in dry acetone. The reaction is known as Finkelstein reaction.
APPEARS IN
संबंधित प्रश्न
Write the major products(s) in the following:
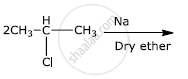
How will you bring about the following conversion?
Benzene to biphenyl
How the following conversion can be carried out?
Benzene to diphenyl
How would you account the following :
Transition metals and their compounds show catalytic properties.
The reaction \[\ce{RX + 2Na + RX -> R - R + 2NaX}\] is called ____________.
Chlorobenzene is prepared commercially by:
Assertion: tert-Butyl bromide undergoes Wurtz reaction to give 2, 2, 3, 3-tetramethylbutane.
Reason: In Wurtz reaction, alkyl halides react with sodium in dry ether to give hydrocarbon containing double the number of carbon atoms present in the halide.
Write balanced equations for a coloured metallic oxide which dissolves in alkalis to yield colourless solutions.
Explain why Grignard reagents should be prepared under anhydrous conditions?
