Advertisements
Advertisements
प्रश्न
Mention the reaction condition and give balanced equation to obtain: Cl2 gas from HCI gas.
उत्तर
\[\ce{2HCl ⇌[>500°C] H2 + Cl2 }\]
APPEARS IN
संबंधित प्रश्न
Identify the gas evolved and give the chemical test in the following cases
Dilute hydrochloric acid reacts with sodium sulphite.
State your observation in given case When dilute hydrochloric acid is added to sodium carbonate crystals
Write the balanced chemical equation for the laboratory preparation of hydrogen chloride gas.
For the preparation of hydrochloric acid in the laboratory:
Why is the direct absorption of hydrogen chloride gas in water not feasible?
Give the balanced equation for the laboratory preparation of hydrogen chloride gas reaction.
Write the main difference in hydrogen chloride gas and hydrochloric acid.
The given set up in the figure is for the preparation of an acid.
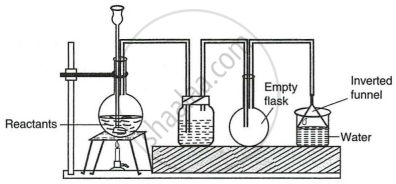
- Name the acid prepared by this method.
- Name the reactants used.
- Why an empty flask is used?
- What is the drying agent used? Why is this drying agent chosen?
- What is the role of the inverted funnel in the arrangement?
Name the gas produced when chlorine water is exposed to sunlight.
Explain, why (or give reasons for)
HCI gas does not conduct electricity, but hydrochloric acid conducts electricity.
What property of hydrogen chloride is demonstrated when it is collected by downward delivery (upward displacement) ?
Hydrogen chloride gas, being highly soluble in water, is dried by ______.
Give one test to distinguish between the following pair of chemicals.
Sodium nitrate solution and sodium chloride solution.
Give one test to distinguish between the following pair of chemicals.
Iron (III) Chloride solution and copper chloride solution.
Answer the following question related to the laboratory preparation of the hydrogen chloride gas:
Why is sodium chloride preferred to other metallic chlorides?
Answer the following question related to the laboratory preparation of the hydrogen chloride gas:
Write the chemical equation.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
