Advertisements
Advertisements
प्रश्न
Name the radiations of wavelength just longer than 8 × 10-7m.
उत्तर
Infrared radiations are longer than 8 × 10-7m = `(8 xx 10^-7)/(10^-10)` = 8000 Å
APPEARS IN
संबंधित प्रश्न
What is the range of the wavelength of the following electromagnetic waves?
(a) Ultraviolet
Can Lα X-ray of one material have shorter wavelength than Kα X-ray of another?
X-ray incident on a material
(a) exerts a force on it
(b) transfers energy to it
(c) transfers momentum to it
(d) transfers impulse to it.
What potential difference should be applied across an X-ray tube to get X-ray of wavelength not less than 0.10 nm? What is the maximum energy of a photon of this X-ray in joule?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name two sources of ultraviolet radiation.
What is the wavelength of the wave whose frequency is 1012 Hz? Name the electromagnetic wave.
The fundamental frequency of an open organ pipe is 300 Hz. The first overtone of this pipe has same frequency as first overtone of a closed organ pipe. If speed of sound is 330 m/s, then the length of closed organ pipe is:
For television broadcasting, the frequency employed is normally
Why does microwave oven heats up a food item containing water molecules most efficiently?
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.
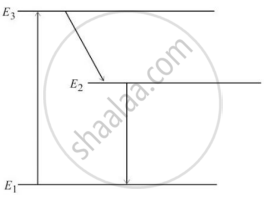
- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
