Advertisements
Advertisements
प्रश्न
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
On the basis of the above reaction, what can you say about the nature of copper oxide?
उत्तर
The reaction between a metal oxide and hydrochloric acid is similar to the neutralisation reaction in which, after the reaction, a salt and water are formed. So, we can conclude that copper oxide is basic in nature.
APPEARS IN
संबंधित प्रश्न
Write the chemical formula of soda ash?
State the common and chemical names of the compound formed when plaster of Paris is mixed with water.
State two uses each of the following compounds:
Sodium hydroxide
Is PbO2 a base or not? Comment.
Phenolphthalein is a synthetic type of indicator.
Choose the correct alternative and rewrite the following sentence.
Phenolphthalein is ___________ type of indicator.
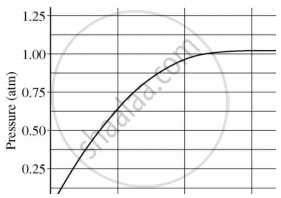
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
What happens when a solution of an acid is mixed with a solution of a base in a test tube?
- The temperature of the solution increases
- The temperature of the solution decreases
- The temperature of the solution remains the same
- Salt formation takes place
Which of the following salts does not contain any water of crystallisation?
Consider the following salt:
YCl
What would be the pH of the solution if, in YCl, Y is sodium? Give a reason for your answer.
