Advertisements
Advertisements
प्रश्न
The atoms of copper contain electrons and the atoms of rubber also contain electrons. Then why does copper conduct electricity but rubber does not conduct electricity?
उत्तर
In a copper atom, the electrons in the outermost orbit are lightly bound to the nucleus of the atom and so they move freely. This free flow of electrons makes copper a good conductor of electricity. In rubber, the electrons in the outermost orbits are tightly bound to the nucleus. So, they do not flow freely and hence, rubber is not a good conductor of electricity.
APPEARS IN
संबंधित प्रश्न
Which instrument is used to measure electric current? How should it be connected in a circuit?
An electric circuit consisting of a 0.5 m long nichrome wire XY, an ammeter, a voltmeter, four cells of 1.5 V each and a plug key was set up.
(i) Draw a diagram of this electric circuit to study the relation between the potential difference maintained between the points 'X' and 'Y' and the electric current flowing through XY.
Which effect of current is utilised in the working of an electric fuse?
A 720 W heater operates from a 220 V line voltage. Calculate:
(a) The electrical energy consumed by the heater in 3 h.
(b) The cost of electrical energy consumption at the rate of 60 paise per kWh.
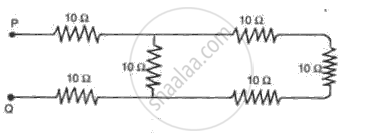
Find the effective resistance in the following circuit diagrams (Fig.):
One unit of electrical energy consumed is equal to 1000 kilowatt-hour.
For current to flow, one needs art open circuit.
