Advertisements
Advertisements
प्रश्न
The diagram given below is of an experiment just at the start. Study the diagram carefully and answer the following questions :
(i) What does the experiment demonstrate?
(ii) Define, the process demonstrated in the experiment.
(iii) What changes are observed after a few hours?
(iv) Give two examples of a semi-permeable membrane.
(v) Which limb of the U-tube contains a more concentrated sucrose solution, A or B?
(vi) Why is the membrane separating the two solutions labeled as semi-permeable membrane?
उत्तर
(i) The process of osmosis.
(ii) Osmosis is a special type of diffusion of solvent molecules through a semi-permeable membrane from a region of their higher concentration to the lower concentration region, but not vice versa.
(iii) The level of water in column A will rise along with the passage of time and will ultimately stop when the concentration of water molecules are equal on both sides of the membrane.
(iv) (a) Parchment paper, (b) Egg membrane.
(v) Solution A.
(vi) It permits the movement of only water molecules to pass through it.
APPEARS IN
संबंधित प्रश्न
What is responsible for guttation?
Give the equivalent terms for the following:
Pressure of the cell contents on the cell wall
Differentiate between the following:
Plasmolysis and deplasmolysis
(a) Mention whether the following statement is true (True) or false (False)
(b) Correct the false statement by altering the last word only.
A plant cell placed in hypotonic solution gets Plasmolysed.
The diagram given below represents an experimental set-up to demonstrate a certain process. Study the same and answer the questions that follow:
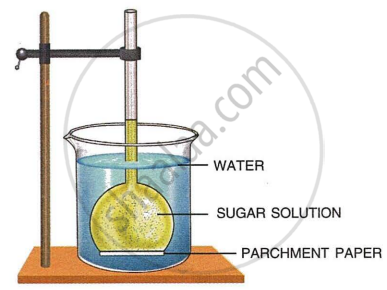
- Name the process.
- Define the above-named process.
- What would you observe in the experimental set-up after an hour or so?
- What control experiment can be set up for comparison?
- Keeping in mind the root hair, cell and its surroundings, name the parts that correspond to
- concentrated sugar solution
- parchment paper
- water in the beaker
- Name any other material that can be used instead of parchment paper in the above experiment.
- Mention two advantages of the process for the plants.
Define the following:
Hypertonic solution
Given below is the figure of an experimental set up to demonstrate root pressure.
(i) Define root pressure.
(ii) What change would you observe in the water level after some time?
(iii) What role is being played by the root pressure in the given experiment?
(iv) Why the oil has been sprinkled on water?
Give Technical Term for the following.
Solutions which have the same osmotic pressure.
Multiple Choice Question:
In the process of osmosis in a cell:
Define the following term:
Osmotic Pressure
