Advertisements
Advertisements
प्रश्न
The number of lone pairs of electrons on the central I atom in `"I"_3^-` is ______.
विकल्प
22
5
3
15
उत्तर
The number of lone pairs of electrons on the central I atom in `"I"_3^-` is 3.
Explanation:
Two nearby I atoms are connected to the central I atom. I atoms have seven valence electrons. The centre I atom will share one electron with each of the two I atoms with which it is linked. On the centre I atom, the final valence electron transforms into lone pairs of electrons.
Iodine is a 7th-group element and a halogen. Therefore, an iodine atom has 7 valence electrons.
The `"I"_3^-` is formed by 3 iodine atoms. There will be one extra electron in `"I"_3^-` which gives it a negative charge.
Therefore, total number of valence electrons in `"I"_3^-` = 7 × 3 + 1 = 22 electrons.
An element atom should have 8 electrons in its valence shell in accordance with the octet rule. The core I atom in I3- contains 8 valence electrons, but there are still 2 more I atoms needed to complete their octet. Since each atom needs eight electrons in its outer shell to complete the octet, both of the nearby iodine atoms will share the one electron that belongs to the centre atom.
Therefore, electron taken by both neighbouring I atoms, 8 - 1 - 1 = 6.
The central I atom has 6 valence electrons which will form 3 lone pairs of electrons which will not involved in bonding. In view of the fact that each iodine atom forms a bond with the central atom, sharing one electron between them, `"I"_3^-` will therefore have three lone pairs of electrons and two bond pairs of electrons.
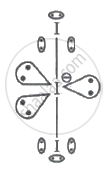
Therefore, the number of lone pairs of electrons on the central I atom in `"I"_3^-` is 3.
