Advertisements
Advertisements
प्रश्न
Toluene reacts with a halogen in the presence of iron (III) chloride giving ortho and para halo compounds. The reaction is ______.
विकल्प
Electrophilic elimination reaction
Electrophilic substitution reaction
Free radical addition reaction
Nucleophilic substitution reaction
उत्तर
Toluene reacts with a halogen in the presence of iron (III) chloride giving ortho and para halo compounds. The reaction is electrophilic substitution reaction.
Explanation:
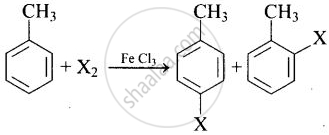
This is an example of electrophilic substitution reaction.
APPEARS IN
संबंधित प्रश्न
How the following conversion can be carried out?
Benzene to 4-bromonitrobenzene
Assertion: Aryl iodides can be prepared by reaction of arenes with iodine in the presence of an oxidising agent.
Reason: Oxidising agent oxidises I2 into HI.
Assertion: In monohaloarenes, further electrophilic substitution occurs at ortho and para positions.
Reason: Halogen atom is a ring deactivator.
Give reason for the following:
During the electrophilic substitution reaction of haloarenes, para-substituted derivative is the major product.
Assertion (A): Chlorobenzene is resistant to electrophilic substitution reaction.
Reason (R): C-Cl bond in chlorobenzene acquires partial double bond characters due to resonance.
Explain why the electrophilic substitution reactions in haloarenes occur slowly and require more drastic conditions as compared to those in benzene.
