Advertisements
Advertisements
प्रश्न
True or false
Dissolving of a solid in liquid is a physical change.
विकल्प
True
False
उत्तर
True
APPEARS IN
संबंधित प्रश्न
Classify the following as chemical or physical change:
Melting of butter in a pan
Choose the most appropriate answer.
Rising and setting of the sun at a given place is a
Write true or false for the statement
Decomposition of leaves is a fast change.
Ripening of fruits is a desirable change.
Solve this crossword by using the clues that follow.
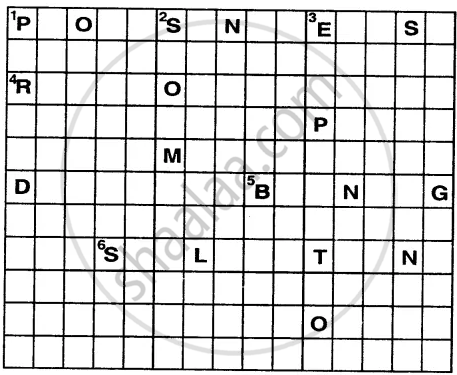
Across
1. Energy in the form of sunlight is absorbed by the green plants in this process.
4. The reddish-brown substance formed over iron in the presence of oxygen and moisture.
5. This change is permanent and irreversible.
6. The process in which a solid directly changes into gaseous state.
Down
1. It is the change that takes place in case of swinging pendulum of a clock.
2. Occurrence of this is a non-periodic change as well as a natural change.
3. It is a physical change
State whether the following is physical or chemical changes.
growing of a piglet into a pig —
Tick the most appropriate answer.
Physical changes are
Which of the following are chemical changes ?
- decaying of wood
- burning of wood
- sawing of wood
- hammering of nail into wood
State which of the following terms connected with change of state of matter i.e.
(a) Melting
(b) Vaporisation
(c) Condensation
(d) Freezing
pertain to –
- Conversion of a liquid into a vapour or gas.
- Conversion of a liquid into a solid.
- Conversion of a solid into a liquid on heating.
Two drops of dilute sulphuric acid were added to 1 g of copper sulphate powder and then a small amount of hot water was added to dissolve it (step I). On cooling, beautiful blue coloured crystals got separated (step II). Step I and Step II are:
