Advertisements
Advertisements
प्रश्न
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:
Ca and O
उत्तर १
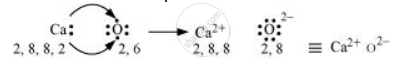
Ca and O:
The electronic configurations of Ca and O are as follows:
Ca: 2, 8, 8, 2
O: 2, 6
Oxygen requires two electrons more to complete its octet, whereas calcium has two electrons more than the nearest noble gas i.e., Argon. Hence, the electron transfer takes place as:
उत्तर २

APPEARS IN
संबंधित प्रश्न
Select and write the most appropriate alternatives from the given choices.
When the following bond types are listed in decreasing order of strength (strongest first). Which is the correct order?
Draw Lewis electron dot structure of HF
Draw Lewis electron dot structure of C2H6
Draw Lewis electron dot structure of C2H4
Draw Lewis electron dot structure of CF3Cl
Draw Lewis electron dot structure of SO2
Explain in brief with one example of a covalent bond
Explain in brief with one example of coordinate bond
Answer in one sentence:
Covalent bond is directional in nature. Justify.
____________ is an example of molecule with expanded octet.
Which of the following is INCORRECT with respect to enthalpy?
In which of the following, the central atom does NOT have lone pair(s) of electrons?
The condensed electronic configuration [Ne] 3s2 3p5 denotes the element ____________.
What is the formal charge on hydrogen atom in water molecule?
Which of the following molecule does not obey octet rule?
