Advertisements
Advertisements
प्रश्न
Using a magnesium ribbon, how would you prove that a given gas jar contains carbon dioxide.
उत्तर
It is a test to find gas in the jar is carbon dioxide. On burning magnesium ribbon in a jar of carbon dioxide.
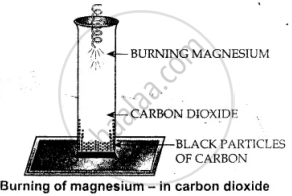
Mg burns in CO2 gas depositing – black particles of carbon on the walls of the jar. If black particles are found on the walls of jar, it is carbon dioxide gas.
CO2 + 2Mg → 2MgO + C
APPEARS IN
संबंधित प्रश्न
Define the following:
Bone black
Name the chemicals required for the preparation of carbon dioxide in the laboratory.
How will you verify the properties of carbon dioxide?
Complete the following chemical reaction.
2NaOH + CO2 `→` ______ + ______
What happens when carbon dioxide is passed through lime water? Write the equation for this reaction.
Lighter metals like Na and K combine with CO2 to form corresponding ______.
What do you mean by aerated water?
Explain the physical properties of carbon dioxide.
