Advertisements
Advertisements
प्रश्न
What is the difference between the molecule of an element and the molecule of a compound ? Give one
example of each.
टिप्पणी लिखिए
उत्तर
| Molecule of an element | Molecule of a compound |
| 1. It consists of two or more atoms of the same element chemically bonded together. | 1. It consists of two or more atoms of different elements chemically bonded together. |
| 2. Example: Bromine exists in nature as a diatomic element (Br2), wherein two bromine atoms are covalently bonded to give a molecule of bromine. | 2. Example: A molecule of nitric acid, HNO3, comprises one atom of hydrogen, one atom of nitrogen and three atoms of oxygen chemically bonded together. |
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
Write down the name of a compound represented by the following formula:
Al2(SO4)3
Define: Atomic number
In what form does oxygen gas occur in nature ?
What do the following denote ?
- N
- 2N
- N2
- 2N2
The cation of an element has :
An element X forms the following compounds with hydrogen, carbon and oxygen :
H2X, CX2, XO2, XO3
State the three valencies of element X which are illustrated by these compounds.
Give one example of a triatomic molecule
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 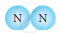 |
|
| 4. | 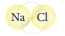 |
Give any two examples for heterodiatomic molecules.
Which of the following contains maximum number of molecules?
