Advertisements
Advertisements
प्रश्न
What happens when a solution of sodium hydrogencarbonate is heated? Write equation of the reaction involved.
उत्तर
When a solution of sodium hydrogen carbonate (NaHCO3) is heated, it decomposes to give sodium carbonate (Na2CO3) with the evolution of carbon dioxide gas.
2NaHCO3 → HeatNa2CO3+ CO2 +H2O +">NaHCOHeat→Na2CO3+CO2+H2O
APPEARS IN
संबंधित प्रश्न
How is the concentration of hydronium ions (H3O+) affected when a solution of an acid is diluted?
What happens when an acid reacts with a metal oxide? Explain with the help of an example. Write a balanced equation for the reaction involved.
A first-aid manual suggests that vinegar should be used to treat wasp stings and baking soda for bee stings. What does this information tell you about the chemical nature of:
bee stings?
Write the chemical formula of sodium carbonate decahydrate.
Complete and balance the following chemical equations:
`Ca(OH)_2 + Cl_2 ->`
State two uses each of the following compounds:
Sodium hydroxide
When _______________ is passed through fresh lime water, it turns milky.
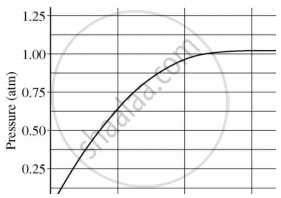
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
A sample of soil is mixed with water and allowed to settle. The clear supernatant solution turns the pH paper yellowish-orange. Which of the following would change the colour of this pH paper to greenish-blue?
Solutions of acids conduct ______.
