Advertisements
Advertisements
प्रश्न
What is the action of following on ethyl bromide?
alcoholic sodium hydroxide
उत्तर
When ethyl bromide is boiled with an alcoholic solution of sodium hydroxide \[\ce{(NaOH)}\], an alkene is formed due to the elimination of hydrogen atoms from carbon and halogen from α-carbon, which is called dehydrohalogenation.
\[\ce{\underset{(Ethyl bromide)}{\overset{\beta\phantom{.............}\alpha\phantom{............}}{CH3 - CH2 - Br}} + \underset{(alc.)}{NaOH} ->[\Delta] \underset{(Ethene)}{CH2 = CH2} + NaBr + H2O}\]
APPEARS IN
संबंधित प्रश्न
from the following pair would undergo SN2 faster from the other?
a. CH3CH2CH2I b. CH3CH2CH2Cl
Name the reagent used to bring about the following conversion.
1-Chloropropane to 1-nitropropane
Give reasons:
Haloarenes are less reactive than haloalkanes.
Convert the following:
Propene to propan-1-ol
Convert the following:
tert-Butyl bromide to isobutyl bromide
The following will react faster by SN1 mechanism
Major product of the following reaction is _______________
\[\ce{CH3-CH2-Mg-Br + NH3 ->?}\]
Nucleophilic substitution reaction of 2, 4-dinitrochlorobenzene is faster than p-nitrochlorobenzene. Give reason.
Explain the factors affecting SN1 and SN2 mechanism.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
Which of the following is a primary halide?
Which of the following is least reactive towards nucleophilic addition?
\[\ce{C6H5CH2Cl + KCN(alc) -> X + Y}\]
Compounds X and Y are ____________.
Identify major product 'B' in the following reaction.
\[\ce{But-1-ene ->[HBr][Peroxide] A ->[AgCN][\Delta] B}\]
Identify 'B' in the following reaction.
\[\ce{2-Bromobutane ->[KOH alc.][\Delta] A ->[HI] B}\]
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
The order of reactivities of the following alkyl halides for an SN2 reaction is ______.
With which halogen the reactions, of alkanes are explosive?
The compound that reacts the fastest with sodium methoxide is ______.
Complete the following reaction giving major products.
\[\begin{array}{cc}
\ce{CH3}\phantom{.......}\\
|\phantom{.........}\\
\ce{CH3 - c - CH2 - Cl ->[Na/dry ether]}\\
|\phantom{.........}\\
\ce{CH3}\phantom{.......}
\end{array}\]
Observe the following and answer the question given below:
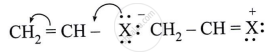
Can react by SN1 mechanism? Justify your answer.
Complete the following reaction, giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Identify the chiral molecule from the following.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Give a reason for the following:
Ethoxy ethane with conc. HI at 373 K gives C2H5OH and CH3I but not CH3OH and C2H5I.
