Advertisements
Advertisements
प्रश्न
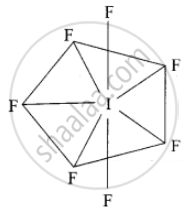
What is the hybridisation of iodine in IF7? Give its structure.
उत्तर
Hybridisation of iodine in IF7 is sp3d3. The structure of IF7 is pentagonal bipyramidal.
APPEARS IN
संबंधित प्रश्न
Assertion: bond dissociation energy of fluorine is greater than chlorine gas
Reason: chlorine has more electronic repulsion than fluorine
Which one of the following compounds is not formed?
Among the following the correct order of acidity is
Give the oxidation state of halogen in the following.
O2F2
What are interhalogen compounds? Give examples.
Give the uses of helium.
Suggest a reason why HF is a weak acid, whereas binary acids of all other halogens are strong acids.
Deduce the oxidation number of oxygen in hypofluorous acid – HOF.
What type of hybridisation occur in BrF3
Complete the following reaction.
\[\ce{HBr + H2SO4 ->}\]
