Advertisements
Advertisements
प्रश्न
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Illustrate the hydroboration-oxidation reaction with an example.
उत्तर
The addition of borane followed by oxidation is known as the hydroboration-oxidation reaction. For example, propan-1-ol is produced by the hydroboration-oxidation reaction of propene. In this reaction, propene reacts with diborane (BH3)2 to form trialkyl borane as an addition product. This addition product is oxidized to alcohol by hydrogen peroxide in the presence of aqueous sodium hydroxide.
\[\ce{\underset{Propene}{CH3 - CH = CH2} + BH3 -> CH3 - CH2 - CH2 - BH2 ->[CH3CH = CH2](CH3 - CH2 - CH2)2BH ->[CH3CH = CH2] \underset{Addition reaction}{(CH3 - CH2 - CH2)3B} ->[H2O/H2O/\overset{-}{O}H]\underset{Propanol}{3CH3 - CH2 - CH2 - OH}}\]
APPEARS IN
संबंधित प्रश्न
Write the main product(s) in each of the following reactions:
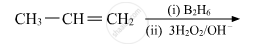
Name the reagents used in the following reactions:
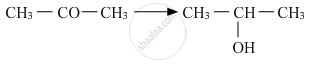
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
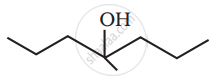
Show how you would synthesise the following alcohol from an appropriate alkene?
Write the structures of A, B and C in the following reactions :
Aldehydes react with Grignard reagent to produce ____________.
Aldehydes are reduced to the corresponding alcohols by the addition of hydrogen in the presence of catalysts to form ____________.
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Ethyl alcohol can be prepared from Grignard reagent by the reaction of ____________.
Commercially carboxylic acids are reduced to alcohols by converting them to the ______.
Explain a process in which a biocatalyst is used in industrial preparation of a compound known to you.
Carboxylic acids are more acidic than phenol and alcohol because of
Most readily hydrolysed halide is:-
When alcohol react with concentrated H2SO4 intermediate compound formed is
Glycerol is used in the manufacture
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide->2-Methylpropan-2-ol}\]
