Advertisements
Advertisements
प्रश्न
What would happen if
Calcium oxide is used to dry hydrogen chloride.
उत्तर
If calcium oxide is used to dry HCI, then it will react with HCI gas and result in the formation of calcium chloride and water.
CaO + 2HCl → CaCl2 + H2O
APPEARS IN
संबंधित प्रश्न
State your observations when Dilute Hydrochloric acid is added to Sodium thiosulphate.
Name the gas that is produced in the given cases :
An action of dilute hydrochloride acid on sodium sulphide.
Name the following:
Gas obtained by treating ferrous sulphide with hydrochloric acid.
Write balanced equation for the following reaction:
Copper oxide and dilute hydrochloric acid.
What is the property of concentrated sulphuric acid which allows it to be used in the preparation of hydrogen chloride and nitric acid?
Write a fully balanced of the following case :
Red lead is warmed with concentrated hydrochloric acid.
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Iron (II) sulphide
Write an equation for the reactions of hydrochloric acid on manganese (IV) oxide.
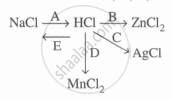
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
