Advertisements
Advertisements
प्रश्न
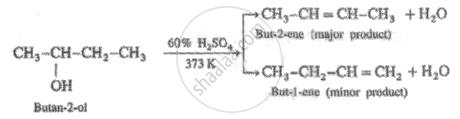
When Butan-2-ol is dehydrated using sulphuric acid, the concentration of acid and temperature needed respectively is _______.
विकल्प
95% cone. and 373 K
60% cone. and 373 K
20% cone. and 363 K
20% cone. and 373 K
MCQ
रिक्त स्थान भरें
उत्तर
When Butan-2-ol is dehydrated using sulphuric acid, the concentration of acid and temperature needed respectively is 60% cone. and 373 K.
Explanation:
Secondary alcohol (2°) is dehydrated by heating with 60% H2SO4 at 373 K.
shaalaa.com
Alkanes
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
