Advertisements
Advertisements
प्रश्न
When a gas jar full of air is placed upside down on a gas jar full of bromine vapours, the red-brown vapours of bromine from the lower jar go upward into the jar containing air. In this experiment :
विकल्प
air is heavier than bromine
both air and bromine have the same density
bromine is heavier than air
bromine cannot be heavier than air because it is going upwards against gravity
उत्तर
Bromine is heavier than air
APPEARS IN
संबंधित प्रश्न
Give a reason for the following observation.
Naphthalene balls disappear with time without leaving any solid.
Give a reason for the following observation.
We can get the smell of perfume sitting several metres away.
Write the full forms of the following
- LPG
- CNG
Explain how, the smell of food being cooked in the kitchen reaches us even from a considerable distance.
Why does a gas fill a vessel completely?
When a gas jar containing colourless air is kept upside down over a gas jar full of brown-coloured bromine vapour, then after some time, the brown colour of bromine vapour spreads into the upper gas jar making both the gas jars appear brown in colour. Which of the following conclusion obtained from these observations is incorrect?
______ is not affected by gravity.
How does the substance of gaseous particles change to a liquid state?
Comment upon the following:-
compressibility
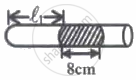 |
|
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)