Advertisements
Advertisements
प्रश्न
Which among the following compounds has highest boiling point?
विकल्प
\[\ce{CH3 - CH2 - CH2 - CH2 - OH}\]
\[\ce{CH3 - CH2 - CH2 - CHO}\]
\[\ce{CH3 - CH2 - COOH}\]
\[\ce{CH3 - CH2 - COCH3}\]
MCQ
उत्तर
\[\ce{CH3 - CH2 - COOH}\]
Explanation:
\[\ce{CH3 - CH2 - COOH}\] compound has the highest boiling point because carboxylic acids are higher boiling liquids than aldehydes. Ketones and even alcohols of comparable molecular masses. This is due to the more extensive association of carboxylic acid molecules through intermolecular hydrogen bonding. The hydrogen bonds are not broken completely even in the vapour phase. In fact, most carboxylic acids exist as a dimer In the vapour phase or in the aprotic solvents.
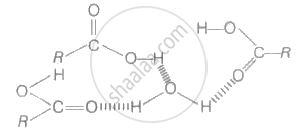
Hydrogen bonding of RCOOH with H2O
shaalaa.com
Aromatic Hydrocarbons
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
