Advertisements
Advertisements
प्रश्न
Which of the following represent the suitable conditions for the liquefaction of gases ?
विकल्प
low temperature, low pressure
high temperature, low pressure
low temperature, high pressure
high temperature, high pressure
उत्तर
low temperature, high pressure
With the help of experiments it can be shown that at low temperature and high pressures, gases will liquefy. Because these factors affect the interaction force of molecules i.e. the molecules are able to establish a force of attraction between them on lowering the temperature and increasing pressure.
APPEARS IN
संबंधित प्रश्न
What is the physical state of water at 250°C.
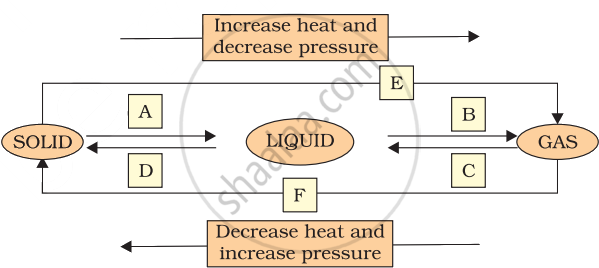
Name A, B, C, D, E and F in the following diagram showing change in its state.
Explain why, a small volume of water in a kettle can fill a kitchen with steam.
What is meant by saying that the latent heat of fusion of ice is 3.34 × 105 J/kg ?
What is the name of the process in which a solid turns directly into a gas ?
When heat is constantly supplied by a gas burner with small flame to melt ice, then the temperature of ice during melting :
The latent heat of fusion of ice is :
The conversion of a solid into vapours without passing through the liquid state is called :
When water is heated to a temperature x, it gets converted into steam at temperature x by a process called R. And when steam at temperature x is cooled, it gets reconverted into water at the same temperature x by a process called S.
- How much is the value of x in Kelvin ?
- What is the process R called ?
- What is the name of the energy absorbed during the process R ?
- What is process S known as ?
- What is the name of energy released during the process S known as ?\\
Fill in the blank with the appropriate word of the statement given below:
Change of milk of curd is a _______ change.
