Advertisements
Advertisements
प्रश्न
Which of the following compounds are gem-dihalides?
(i) Ethylidene chloride
(ii) Ethylene dichloride
(iii) Methylene chloride
(iv) Benzyl chloride
उत्तर
(i) Ethylidene chloride
(iii) Methylene chloride
Explanation:
Gem-dihalides are those dihalides in which two halogen atoms are bonded to the same carbon atom.
(i) \[\ce{\underset{Ethylidene chloride}{Cl2 - CH - CH3}}\]
(ii) \[\ce{\underset{Ethylene dichloride}{Cl - H2C - CH2 - Cl}}\]
(iii) \[\ce{\underset{Methylene chloride}{CH2Cl2}}\]
(iv)
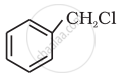
Benzyl chloride
So, in (i) and (iii) two halogen atoms are present on the same carbon atom and they are termed gem-dihalides.
APPEARS IN
संबंधित प्रश्न
Write the structure of the following compound:
4-tert. Butyl-3-iodoheptane
Write structures of different dihalogen derivatives of propane.
Write the structure of the following organic halogen compound.
p-Bromochlorobenzene
Among the following which one can have a meso form?
Benzene diazonium chloride reacts with hypophosphorous acid to produce ____________.
Give the IUPAC names of the following compound:
(CCl3)3CCl
Give the IUPAC name of the following compound:
CICH2C ≡ CCH2Br
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following compound:
4-tert-Butyl-3-iodoheptane
Write the structure of the following compound:
1,4-Dibromobut-2-ene
