Advertisements
Advertisements
प्रश्न
Which of the following is CORRECT?
\[\ce{H3PO4_{(aq)} + 3KOH_{(aq)} -> K3PO4_{(aq)} + 3H2O_{(l)}}\]
विकल्प
Oxidation state of P changes from +5 to +6.
Except "P", oxidation state of all other elements remains constant.
The oxidation numbers of all the species remain same.
"P" acts as a reductant whereas "O" acts as oxidant.
MCQ
उत्तर
The oxidation numbers of all the species remain same.
Explanation:
Given reaction,
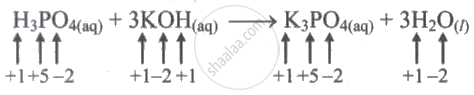
This is an example of an acid-base reaction. Because all of the species' oxidation values are the same, the reaction is not a redox reaction.
shaalaa.com
Oxidation Number
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
