Advertisements
Advertisements
प्रश्न
Which of the following polymers, need atleast one diene monomer for their preparation?
(i) Dacron
(ii) Buna-S
(iii) Neoprene
(iv) Novolac
उत्तर
(ii) Buna-S
(iii) Neoprene
Explanation:
\[\begin{array}{cc}\phantom{}
\phantom{..................................................................}\ce{C6H5}\\
\phantom{...............................................................}|\\
\ce{\underset{1, 3-Butadiene}{nCH2 = CH} - CH = CH2 + n C6\underset{Styrene}{H5CHCH2} -> \underset{(Buna - S)}{\underset{Butadiene-styrene copolymer}{-(CH2 - CH = CH - CH2 - CH - CH2)\underset{n}{-}}}}
\end{array}\]
Neoprene or polychloroprene is formed by the free radical polymerisation of chloroprene.
\[\begin{array}{cc}
\phantom{..}\ce{Cl}\phantom{...................................}\ce{Cl}\phantom{.........}\\
\phantom{}|\phantom{.....................................}|\phantom{........}\\
\ce{\underset{2-Chloro-1, 3-buta-1, 3-diene}{\underset{Chloroprene}{n CH2 = C - CH = CH2}} ->[Polymerisation] \underset{Neoprene}{-[CH2 - C = CH - CH2]\underset{n}{-}}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Explain the following term: Homopolymers
Based on molecular forces, what type of polymer is neoprene?
Answer the following in one sentence.
Identify 'A' in the following reaction:
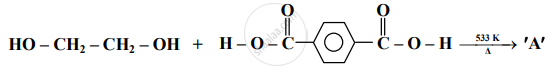
Identify additional polymers from the following.
I. \[\begin{array}{cc}
\ce{-(CH2 - CH -)_{{n}}}\\
\phantom{....}|\\
\phantom{.......}\ce{C6H5}
\end{array}\]
II. \[\ce{-(CH2 - CH = CH - CH2 -)_{{n}}}\]
III. \[\ce{-(CO(CH2)4 - CONH(CH2)6NH -)_{{n}}}\]
IV.
![]()
Which of the following polymers is a heteropolymer?
Which among the following polymers can NOT be remoulded?
Which of the following is not a semisynthetic polymer?
Which of the following is not a copolymer?
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
