Advertisements
Advertisements
प्रश्न
Which of the following solutions have the same concentration?
(i) \[\ce{20 g of NaOH in 200 mL of solution}\]
(ii) \[\ce{0.5 mol of KCl in 200 mL of solution}\]
(iii) \[\ce{40 g of NaOH in 100 mL of solution}\]
(iv) \[\ce{20 g of KOH in 200 mL of solution}\]
उत्तर
(i) \[\ce{20 g of NaOH in 200 mL of solution}\]
(ii) \[\ce{0.5 mol of KCl in 200 mL of solution}\]
Explanation:
(i) The number of moles is given by the following formula,
Moles = `"Mass"/"Molar mass"` ......(1)
So, the number of moles of \[\ce{NaOH}\] is calculated, by using equation (1) as follows,
Moles of NaOH = `(20 g)/((40 g)/(mol)` = 0.5 mol
The molarity (M) is given by the formula:
M = `n/V_((L))`
On substituting the values in the above equation:
MNaCH = `(0.5 mol xx 1000)/(200 L)` = 2.5 mol L–1
(ii) The molarity is given by the formula:
M = `n/V_((L))`
On substituting the values in the above equation:
MKCl = `(0.5 mol xx 1000)/(200 L)` = 2.5 mol L–1
APPEARS IN
संबंधित प्रश्न
Calculate the amount of carbon dioxide that could be produced when 1 mole of carbon is burnt in air.
To make a saturated solution, 36 g of sodium chloride is dissolved in 100 g of water at 293 K. Find its concentration at this temperature.
Solve the following problem:
Find out the molar mass of the following compounds:
Sodium carbonate, decahydrate (Na2CO3.10H2O)
(At. mass: Cu = 63.5; S = 32; O = 16; H = 1; Na = 23; C = 12; Fe = 56; N = 14)
Solve the following problem:
Write the following number in ordinary decimal form:
5.16 × 104
Solve the following problem:
Write the following number in ordinary decimal form:
43.71 × 10−4
Solve the following problem:
Write the following number in ordinary decimal form:
14.3 × 10−2
Solve the following problem:
Perform the following calculation. Round off your answer to two digits.
`33/(9.00xx10^-4)`
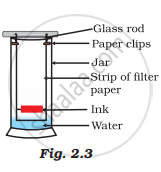
A child wanted to separate the mixture of dyes constituting a sample of ink. He marked a line by the ink on the filter paper and placed the filter paper in a glass containing water as shown in Fig.2.3. The filter paper was removed when the water moved near the top of the filter paper.
(i) What would you expect to see, if the ink contains three different coloured components?
(ii) Name the technique used by the child.
(iii) Suggest one more application of this technique.
With increase of temperature, which of these changes?
250 g solution of D-glucose in water contains 10.8% of carbon by weight. The molality of the solution is nearest to ______.
(Given: Atomic weights are H, 1u; C, 12u; O, 16u)
