Advertisements
Advertisements
प्रश्न
Why does our palm feel cold when we put some acetone (or perfume) on it ?
उत्तर
Our palm feels cool when we put acetone over it because acetone takes up the latent heat of vaporisation to change into vapour from our palm.
APPEARS IN
संबंधित प्रश्न
What type of clothes should we wear in summer?
Explain with an example to demonstrates that when a liquid evaporates, it takes heat from its surroundings.
Would you cool a bucket of water more quickly by placing it on ice or by placing ice in it ? Give reasons for your answer.
If the back of your hand is moistened with alcohol, you will find that it rapidly becomes dry. Why is it that while it is drying, your hand feels cool ?
How can the evaporation of a liquid be made faster ?
Why is the air in a region dry?
How is humidity measured?
Evaporation of water from lakes and river ______.
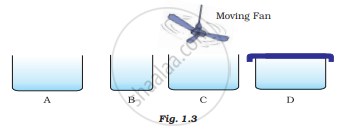
Look at Fig. 1.3 and suggest in which of the vessels A,B, C or D the rate of evaporation will be the highest? Explain.
The rate of evaporation ______ with rising temperature.
