Advertisements
Advertisements
प्रश्न
Why is ice at 273 K more effective in cooling than water at the same temperature?
उत्तर
- Ice at 273 K has less energy than water (although both are at the same temperature).
- Water possesses the additional latent heat of fusion. Hence, at 273 K, ice is more effective in cooling than water.
APPEARS IN
संबंधित प्रश्न
What is the physical state of water at 0°C.
What is the (a) common unit of temperature, and (b) SI unit of temperature ?
How is ammonia gas liquefied ?
When heat is constantly supplied by a gas burner with small flame to melt ice, then the temperature of ice during melting :
The latent heat of fusion of ice is :
Which of the following energy is absorbed during the change of state of a substance ?
What is it made from? Why?
A sickle to cut sugarcane.
Who am I?
I have no shape whatsoever!
Complete the blank with reference to interconversion of matter on the basis of kinetic theory-with the word ‘increase’, ‘decreases’, ‘gain’, ‘lose’, or ‘overcome’ in the following case.
During vaporization, the liquid particles ____________ energy.
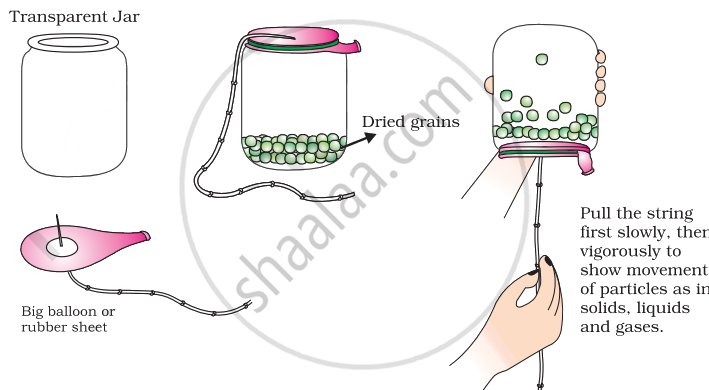
Prepare a model to demonstrate movement of particles in solids, liquids and gases.
For making this model you will need
- A transparent jar
- A big rubber balloon or piece of stretchable rubber sheet
- A string
- Few chickpeas or black gram or dry green peas.
How to make?
- Put the seeds in the jar.
- Sew the string to the centre of the rubber sheet and put some tape to keep it tied securely.
- Stretch and tie the rubber sheet on the mouth of the jar.
- Your model is ready. Now run your fingers up and down the string by first tugging at it slowly and then rapidly.