Advertisements
Advertisements
प्रश्न
Write IUPAC name of the product ‘B’ in the following reaction sequence.
\[\ce{C2H5OH ->[NaBr/H2SO4][\Delta] A ->[Nal][acetone]B}\]
उत्तर
The IUPAC name of the product ‘B’ formed in the following reaction sequence is iodoethane.
\[\ce{\underset{\text{Ethanol}}{C2H5 - OH} ->[NaBr,H2SO4][heat] \underset{\text{Bromoethane (A)}}{C2H5 - Br} ->[Nal][Acetone] \underset{\text{Iodoethane (B)}}{C2H5 - I} + NaBr↓}\]
APPEARS IN
संबंधित प्रश्न
An alkyl chloride on Wurtz reaction gives 2,2,5,5-tetramethylhexane. The same alkyl chloride on reduction with a zinc-copper couple in alcohol gives hydrocarbon with molecular formula \[\ce{C5H12}\]. What is the structure of alkyl chloride?
Choose the most correct option.
Which of the following is used as a source of dichlorocarbene?
Write the IUPAC name of the following.
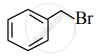
Write the major product of the following reaction.
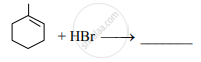
How are the following conversions carried out?
propene to 1-iodopropane
How are the following conversions carried out?
propene to 2-nitropropane
How are the following conversions carried out?
benzene to biphenyl
Write chemical equations indicating the action of following on bromobenzene.
CH3COCl/anhy. AlCl3
Write chemical equations indicating the action of following on bromobenzene.
conc. HNO3/conc. H2SO4
Write the equations for the preparation of ethyl chloride using
- Hydrogen halide
- ethene
- Thionyl chloride
Which of these methods is preferred and why?
Lucas reagent is a mixture of ____________.
Identify 'B' in the following reaction.
\[\ce{C2H6 ->[Br2][AlBr3] A ->[CH3COOAg][\Delta] B}\]
Which of the following reactions does NOT occur?
When one mole of [Co(NH3)5Cl]Cl2 is treated with excess of AgNO3 solution, ____________ mole(s) of AgCl(s) is formed.
The number of asymmetric carbon atoms present in 2, 3-dichloro-4-methyl pentane is ____________.
Phosgene is the common name for ______.
Freon used as refrigerant is ______.
The gas(es) not responsible for ozone depletion:
End product of the following sequence of reactions is:

Why chloroform is stored in dark coloured air tight bottles?
