Advertisements
Advertisements
प्रश्न
Write the molecular formula of the 2nd and the 3rd member of the homologous series whose first member is methane.
उत्तर
The general formula for the homologous series whose first member is methane is CnH2n+2.
The two consecutive members (2nd and 3rd) of this series are:
a. CH3CH3 (Ethane); where n is 2
b. CH3CH2CH3(Propane); where n is 3
The molecular formulae are C2H6 and C3H8 respectively.
APPEARS IN
संबंधित प्रश्न
Give a balanced chemical equation for Action of alcoholic KOH on bromethane
Identify the term/substance in the following:
The catalyst used in the conversion of ethyne to ethane.
Organic compounds having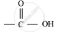 functional group are known as
functional group are known as
Three organic compounds A, B and C have the following molecular formulae: C4H8O2
Which compound contains an alcohol group? Write its name and structural formula.
Give three uses of ethanol
Write the structural formulae of the following compound:
Ethane
Which of the following are used as anesthetics?
A gas is evolved when ethanol reacts with sodium. Name the gas evolved and also write the balanced chemical equation of the reaction involved.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
