Advertisements
Advertisements
प्रश्न
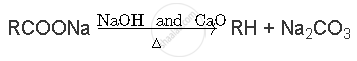
Write the reactions involved Decarboxylation reaction
उत्तर
Decarboxylation reaction: Carboxylic acid lose CO2 to form hydrocarbons when their sodium salts are heated with sodalime (NaOH and CaO in the ratio 3:1). The reaction is known as decarboxylation reaction
APPEARS IN
संबंधित प्रश्न
Predict the products of the following reactions :
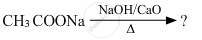
How will you bring about the following conversion in not more than two steps?
Benzoic acid to Benzaldehyde
Describe the following:
Decarboxylation
Give a reason for the following :
N-N bond is weaker than the P-P bond.
Which of the following is the strongest acid?
Which will undergo decarboxylation rapidly?
Maximum decarboxylation occurs in:
Identify product A in the following reaction.
\[\begin{array}{cc}
\ce{COOH}\phantom{.......}\\
/\phantom{...............}\\
\ce{CH2 ->[\Delta] CH3COOH + A}\\
\backslash\phantom{................}\\
\ce{COOH}\phantom{........}
\end{array}\]
Give the order of decarboxylation of the following acid:
| CH3COOH | CH2 = CH–CH2 – COOH |
| I | II |
| CH2(COOH)2 | 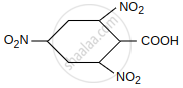 |
| III | IV |
