Advertisements
Advertisements
प्रश्न
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
Zn − Hg/conc. HCl
उत्तर
Acetone undergoes Clemmensen reduction in presence of zinc amalgam and conc. HCl to give propane
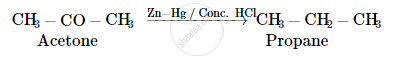
संबंधित प्रश्न
How will you bring about the following conversions?
Propanone to propane
Predict the products of the following reactions :
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Zinc amalgam and dilute hydrochloric acid
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{CH3COCH2COOC2H5 ->[(i) NaBH4][(ii) H+]}\]
Predict the products of the following reactions :
Predict the main product of the following reaction:\[\begin{array}{c}
\ce{O\phantom{----}O\phantom{-}}\\
\ce{||\phantom{----}||\phantom{-}}\\
\ce{CH3-C-CH2-C-OCH3}
\end{array}\ce{->[(i)NaBH4][(ii)H+]}\]
Write chemical equation of the following reaction :
Acetophenone is treated with `("Zn"("Hg"))/"Conc.HCl"`.
Reduction of > C = O to CH2 can be carried out with
Explain the following reactions:
Clemmensen reaction
Write the main product in the following reaction:
\[\ce{CH3CH2CHO ->[Zn(Hg)/Conc. HCl]}\]
