Advertisements
Advertisements
प्रश्न
Write the chemical formula of the following compound in a step-by-step manner –
Iron [II] hydroxide
उत्तर
Write the formula of
| Iron [II] hydroxide | Symbol | Valency |
| Iron | \[\ce{Fe}\] | \[\ce{2^{+}}\] |
| Hydroxide | \[\ce{OH}\] | \[\ce{1^{-}}\] |
Step I – Write each symbol with its valency.
Positive ion is written first
\[\ce{Fe}^{2+}\] \[\ce{[OH]}^{1-}\]
Step II – Interchange the valencies ignoring signs.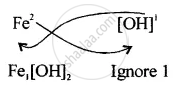
The Formula is \[\ce{Fe[OH]2}\]
APPEARS IN
संबंधित प्रश्न
Write the molecular formula for the oxide and sulphide of following elements of Hydrogen
Write the molecular formulae of Iron (II) sulphide.
Write the molecular formulae of Hydrogen sulphate (sulphuric acid).
Write the molecular formulae of Calcium hydroxide.
Explain the term ‘variable valency’.
Write the chemical formula of the following compound in a step-by-step manner –
Sodium bromide
Write the chemical formula of the following compound in a step-by-step manner –
Sodium zincate
Write the chemical formula of the following compound in a step-by-step manner –
Copper [I] sulphide
Write the chemical formula of the following compound in a step-by-step manner –
Iron [III] chloride
Complete the statement given below by filling in the blank with the correct word.
The formula of silver [I] chloride is ________.
