Advertisements
Advertisements
प्रश्न
Write the first four members of the homologous series that begins with H-C≡C-H. Also, write down their general molecular formula.
उत्तर
| No. | Name of a compound and molecular formula | Structural formula |
| 1. | Ethyne (C2H2) | HC ≡ CH |
| 2. | Propyne (C3H4) | CH3 - C ≡ CH |
| 3. | But-1-yne (C4H6) | CH3 - CH2 - C ≡ CH |
| 4. | Pent-1-yne (C5H8) | CH3 - CH2 - CH2 - C ≡ CH |
Comparing these molecular formulae and assigning the number of carbon atoms as ‘n’, the following general formula is deduced: CnH2n - 2 (where n = 2, 3, 4...)
APPEARS IN
संबंधित प्रश्न
Draw a formula for the first five members of the homologous series beginning with the given compound.
CH3COCH3
Draw a formula for the first five members of the homologous series beginning with the given compound.
H–CH=CH2
Identify the functional group in the following compound.

Identify the functional group in the following compound.
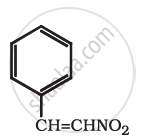
Find out all the functional groups present in the following polyfunctional compound.
Thyroxine, the principal thyroid hormone.
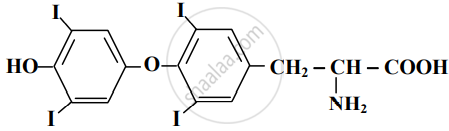
Write the first four members of the homologous series that begins with CH3CHO. Also, write down their general molecular formula.
How many cyclic and acyclic isomers are possible for the molecular formula C3H6O?
Write the molecular formula and possible structural formula of the first four members of homologous series of carboxylic acids.
Write the molecular formula of the first six members of homologous series of nitro – alkanes.
What is the percentage of carbon in urea?
(Atomic mass C = 12, H = 1, N = 14, O = 16)
Which of the following yields only one type of monosubstituted chloroalkane upon chlorination?
Which of the following is NOT a cyclic compound?
How many 3° carbon atoms are present in 1, 4 dibromo-2-methylpentane?
What is the molecular formula of pyran?
How many primary, secondary and tertiary carbon atoms respectively are present in isobutane?
Identify the number of carbon atoms and number of oxygen atoms respectively present in furan molecule.
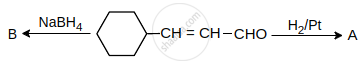
A and B are:
Identify primary, secondary, tertiary, and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3} \ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
\phantom{}|\phantom{....}|\phantom{...............}\\
\phantom{}\ce{CH3}\phantom{.}\ce{CH3}\phantom{.............}
\end{array}\]
Identify the α - carbons in the following species and give the total number of
\[\ce{CH3 -CH2 -\overset{⊕}{C}H -CH2 -CH3}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\ce{CH3}\phantom{..............}
\end{array}
Indicate the σ and π bonds in the following molecule:
\[\ce{HCONHCH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{.....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
