Advertisements
Advertisements
प्रश्न
Write the formula of the compound formed – given symbols and combining power or capacity (valency) of the following element in the compound.
K1+ Cl1-
उत्तर
K1+ Cl1-
Steps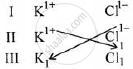
Formula = KCl
संबंधित प्रश्न
The atoms of which of the following pair of elements are most likely to exist in free state ?
The formula of the sulphate of an element X is X2(SO4)3. The formula of nitride of element X will be :
A student wants to have 3.011 × 1023 atoms each of magnesium and carbon elements. For this purpose, he will have to weigh :
The formula of the sulphate of an element M is M2(SO4)3
Write the formula of it
Oxide
Match the symbols of – radicals – with their correct combining power or capacity, (valency).
(a) OH
(b) So4
(c) No3
(d) Co3
Combining power or capacity – A : 3; B : 2; C : 1. (negative valencies)
Give the symbol of the following element:
Calcium
Give the formula of the following acid:
Sulphuric
Give the formula of the following gas:
Hydrogen chloride
State whether the following ion or radical given below of a non-metallic element exhibits valency: 1–, 2– or 3–. (write the ion or radical with the correct valency).
Nitride
What does a chemical symbol represent?
