Advertisements
Advertisements
प्रश्न
Write the molecular formula and structural formula for the following molecule.
Nitric acid
सारिणी
उत्तर
| Molecule | Molecular formula | Structural formula |
| Nitric acid | HNO3 | 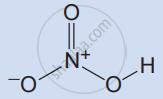 |
shaalaa.com
Group 15 (Nitrogen Group) Elements
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
In which of the following, NH3 is not used?
Which is true regarding nitrogen?
An element belongs to group 15 and 3rd period of the periodic table, its electronic configuration would be
P4O6 reacts with cold water to give ____________.
The molarity of given orthophosphoric acid solution is 2M. its normality is ____________.
Write the molecular formula and structural formula for the following molecule.
Phosphine
Write the valence shell electronic configuration of group-15 elements.
Give a reaction between nitric acid and a basic oxide.
Complete the following reaction.
\[\ce{NaNO2 + HCl ->}\]
Complete the following reaction.
\[\ce{KClO3 ->[\Delta]}\]
