Advertisements
Advertisements
प्रश्न
Write the molecular formula of the given compound.
Sodium ethoxide
उत्तर
Sodium ethoxide- C2H5ONa
APPEARS IN
संबंधित प्रश्न
How can ethanol and ethanoic acid be differentiated on the basis of their physical properties?
Give the structural formula of Ethanol
Draw the structures for the following compounds of Pentanal
Complete the following equation:
`CH_3 CH_2 OH` 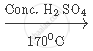
What is meant by denatured alcohol? What is the need to denature alcohol?
How would you convert of thanol into ethene?
How is the absolute alcohol obtained?
How is the spurious alcohol obtained?
What would you see, when ethyne is bubbled through a solution of bromine in carbon tetrachloride?
Identify the functional group of the CH3OH ?
What is meant by functional group in carbon compounds? Write in tabular form the structural formula and the functional group present in the following compounds:
(i) Ethanol
(ii) Ethanoic acid
Ethanol can be converted to ethene which can be changed to ethane.
Choose the correct word or phrase from the options given below to complete the following sentences:
Converting ethanol to ethene requires the use of_________(Concentrated hydrochloric acid, concentrated nitric cid, concentrated sulphuric acid).
Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol
From the compounds given above, name
The compound with -OH as apart of its structure.
[Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol]
From the above list, name the compound with \[\ce{-OH}\] as the part of its structure.
How would you convert ethene into ethane?
Write the molecular formula of the given compound.
Ethanol
Colorless ethanol is in liquid state at room temperature.
Ethene is formed when ethanol at 443 K is heated with excess of concentrated sulphuric acid. What is the role of sulphuric acid in this reaction? Write the balanced chemical equation of this reaction.
What is the role of concentrated Sulphuric acid when it is heated with Ethanol at 443 K. Give the reaction involved.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
