Advertisements
Advertisements
प्रश्न
Write the Van der Waals equation for a real gas. Explain the correction term for pressure and volume.
उत्तर
Vander Waals equation for a real gas is given by
`("P" + ("an"^2)/("V"^2)) ("V" - "nb") = "nRT"`
Pressure Correction:
The pressure of a gas is directly proportional to the force created by the bombardment of molecules on the walls of the container. The speed of a molecule moving towards the wall of the container is reduced by the attractive forces exerted by its neighbours. Hence, the measured gas pressure is lower than the ideal pressure of the gas. Hence, van der Waals introduced a correction term to this effect.
Where n is the number of moles of gas and V is the volume of the container
`"p" ∝ "n"^2/"V"^2`
`"P" = "an"^2/"V"^2`
Where a is proportionality constant and depends on the nature of gas
Therefore, P = `"P" + "an"^2/"V"^2`
Volume Correction:
As every individual molecule of a gas occupies a certain volume, the actual volume is less than the volume of the container, V. Van der Waals introduced a correction factor V to this effect. Let us calculate the correction term by considering gas molecules as spheres.
V = excluded volume
Excluded volume for two molecules = `4/3` π(2r)3 = 8Vm
where Vm is a volume of a single molecule.
Excluded volume for single molecule = `(8"V"_m")/2` = 4Vm
Excluded volume for n molecule = n(4Vm) = nb
Where b is van der Waals constant which is equal to 4Vm
V’ = nb
Videal = V – nb
Replacing the corrected pressure and volume in the ideal gas equation PV = nRT we get the van der Waals equation of state for real gases as below,
`("P" + ("an"^2)/("V"^2)) ("V" - "nb") = "nRT"`
The constants a and b are van der Waals constants and their values vary with the nature of the gas.
APPEARS IN
संबंधित प्रश्न
Calculate the volume occupied by 8.8 g of CO2 at 31.1°C and 1 bar pressure. R = 0.083 bar L K–1 mol–1.
Compressibility factor for CO2 at 400 K and 71.0 bar is 0.8697. The molar volume of CO2 under these conditions is
Maximum deviation from ideal gas is expected from
Which of the following diagrams correctly describes the behaviour of a fixed mass of an ideal gas? (T is measured in K)
25 g of each of the following gases are taken at 27°C and 600 mm Hg pressure. Which of these will have the least volume?
Explain whether a gas approaches ideal behavior or deviates from ideal behaviour if more gas is introduced into the same volume and at the same temperature.
Under which of the following two conditions applied together, a gas deviates most from the ideal behaviour?
(i) Low pressure
(ii) High pressure
(iii) Low temperature
(iv) High temperature
Compressibility factor, Z, of a gas is given as Z = `(pV)/(nRT)`. What is the value of Z for an ideal gas?
Match the following graphs of ideal gas with their co-ordinates:
| Graphical representation | x and y co-ordinates |
(i)  |
(a) pV vs. V |
(ii) 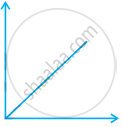 |
(b) p vs. V |
(iii) 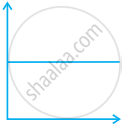 |
(c) p vs. `1/V` |
In van der Waal's equation for the real gas, the expression for the net force of attraction amongst the gas molecules is given by:
