Advertisements
Advertisements
प्रश्न
Write three uses of sulfuric acid.
उत्तर
Sulfuric acid is a very important industrial chemical.
It is used:
- in the manufacture of fertilizers. For example, ammonium sulfate, superphosphate, etc.
- in the manufacture of pigments, paints and dyestuff intermediates.
- in petroleum refining.
- in detergent industry.
- in metallurgy, for cleaning of metals electroplating and galvanising.
- in storage batteries.
- as a laboratory reagent.
- in the manufacture of nitrocellulose products.
संबंधित प्रश्न
Answer the following.
What is the oxidation state of ‘S’ in H2SO4?
Answer the following.
What happens when Br2 reacts with excess of F2.
Write a balanced chemical reaction of sulfuric acid with sulfur.
What is oxidation state of sulfur in the following?
Peroxymonosulfuric acid
Write three physical properties of sulfuric acid.
Draw the structure of chloric acid.
The formula of chlorous acid is ____________.
The molecular formula of metaperiodic acid is ____________.
Which of the following oxyacids of sulphur contain four S = 0 bonds?
Which of the following molecular formula represents Marshall's acid?
The following structure represents
Which of the following structure represents thiosulfuric acid?
The number of S = O bond in peroxy monosulfuric acid is _____________.
The oxidation number of sulfur in the product is ____________.
\[\ce{I2 + SO2 + 2H2O ->}\]
Which of the following structure represents chloric acid?
Which of the following oxyacid of sulphur contain both S=S and S=O bonds?
Identify the correct decreasing order of oxidizing power.
What is the oxidation state of chlorine atom in hypochlorous acid?
How many lone pairs of electrons are present on each oxygen atom in any oxyacids of chlorine?
How many lone pair of electrons are present on chlorine atoms in Hypochlorous acid?
Which among the following oxoacids of phosphorus shows a tendency of disproportionation?
Draw the structure of oxyacid of sulphur in which the oxidation state of sulphur is + 4.
How is chlorine obtained by oxidation of HCl. Write the reactions using two oxidizing agents.
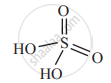
Draw structure of H2SO4.
Draw the structure of peroxymonosulphuric acid.
