Advertisements
Advertisements
प्रश्न
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with iron filings.
उत्तर १
- \[\ce{Iron filings + Hydrochloric acid (dil{.}) -> Iron (II) chloride + Hydrogen}\]
- \[\ce{Fe_{(s)} + 2HCl(dil{.}) -> FeCl2_{(aq)} + H2_{(g)}}\]
उत्तर २
- \[\ce{Hydrochloric acid + Iron -> Iron(III) chloride + Hydrogen}\]
- \[\ce{6HCl_{(aq)} + 2Fe_{(s)} −> 2FeCl3_{(aq)} + 3H2_{(g)}}\]
APPEARS IN
संबंधित प्रश्न
| Column A | Column B | ||
| i | eosin | 1 | losing hydrogen |
| ii | oxidation | 2 | synthetic indicator |
| 3 | losing oxygen | ||
| 4 | natural indicator |
From which plant is litmus paper or litmus solution obtained?
- Moss
- Rose
- Hibiscus
- Lichen
Why do HCl, HNO3, etc., show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with magnesium ribbon.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute sulphuric acid reacts with aluminium powder.
What is a neutralisation reaction?
A first-aid manual suggests that vinegar should be used to treat wasp stings and baking soda for bee stings. What does this information tell you about the chemical nature of:
wasp stings?
A first-aid manual suggests that vinegar should be used to treat wasp stings and baking soda for bee stings. What does this information tell you about the chemical nature of:
bee stings?
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Write the chemical equation of the reaction of metal X with (i) hydrochloric acid, and (ii) sodium hydroxide solution.
State two uses each of the following compounds:
Sodium hydroxide
Magnesium hydroxides are used for treating _______
What happens when a solution of an acid is mixed with a solution of a base in a test tube?
- The temperature of the solution increases
- The temperature of the solution decreases
- The temperature of the solution remains the same
- Salt formation takes place
Which of the following salts does not contain any water of crystallisation?
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
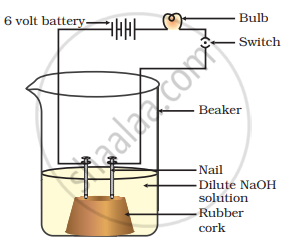
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
Name the acid present in ant sting and give its chemical formula. Also give the common method to get relief from the discomfort caused by the ant sting.
What happens when nitric acid is added to egg shell?
In the following schematic diagram for the preparation of hydrogen gas as shown in the figure, what would happen if following changes are made?

- In place of zinc granules, same amount of zinc dust is taken in the test tube
- Instead of dilute sulphuric acid, dilute hydrochloric acid is taken
- In place of zinc, copper turnings are taken
- Sodium hydroxide is taken in place of dilute sulphuric acid and the tube is heated.
Vinegar is ______ in taste.
“All acids contain one or more hydrogen atoms, but not all substances containing hydrogen are acids.” Support this statement with examples.
