Advertisements
Advertisements
Give reasons Conductivity of CH3COOH decreases on dilution.
Concept: Conductance of Electrolytic Solutions > Measurement of the Conductivity of Ionic Solutions
For the reaction
`2AgCl (s) + H_2 (g) ("1 atm") -> 2Ag (s) + 2H^(+) (0.1 M) + 2Cl^(-) (0.1 M)`
`triangleG^0 = -43600 J at 25^@ C`
Calculate the e.m.f. of the cell
`[log 10^(-n) = -n]`
Concept: Relation Between Gibbs Energy Change and Emf of a Cell
An analysis shows that FeO has a non-stoichiometric composition with formula Fe0.95O. Give reason.
Concept: General Properties of the Transition Elements (D-block)
Complete and balance the following chemical equations
`Fe^(2+) + MnO_4^(-) + H^+ ->`
Concept: General Properties of the Transition Elements (D-block)
Complete and balance the following chemical equations
`MnO_4^(-) + H_2O + I^(-) ->`
Concept: General Properties of the Transition Elements (D-block)
Give reasons: E° value for the Mn3+/Mn2+ couple is much more positive than that for Fe3+/Fe2+.
Concept: General Properties of the Transition Elements (D-block)
Give reasons Iron has the higher enthalpy of atomization than that of copper.
Concept: General Properties of the Transition Elements (D-block)
Give reasons: Sc3+ is colourless in aqueous solution whereas Ti3+ is coloured.
Concept: General Properties of the Transition Elements (D-block)
Write the formula for the given coordination compound:
Iron (III) hexacyanoferrate (II)
Concept: Introduction of Coordination Compounds
CO (g) and H2 (g) react to give different products in the presence of different catalysts. Which ability of the catalyst is shown by these reactions?
Concept: Catalysis > Catalysts in Industry
Write the coordination number and oxidation state of Platinum in the complex [Pt(en)2Cl2].
Concept: Coordination Compounds Numerical
What happens when a freshly prepared precipitate of Fe(OH)3 is shaken with a small amount of FeCl3 solution?
Concept: Properties of Colloidal Solutions
What happens when persistent dialysis of a colloidal solution is carried out?
Concept: Properties of Colloidal Solutions
What happens when an emulsion is centrifuged?
Concept: Properties of Colloidal Solutions
(A), (B) and (C) are three non-cyclic functional isomers of a carbonyl compound with molecular formula C4H8O. Isomers (A) and (C) give positive Tollens' test whereas isomer (B) does not give Tollens' test but gives positive Iodoform test. Isomers (A) and (B) on reduction with Zn(Hg)/conc. HCl give the same product (D).
1) Write the structures of (A), (B), (C) and (D).
2) Out of (A), (B), and (C) isomers, which one is least reactive towards the addition of HCN?
Concept: Bonding in Metal Carbonyls
Write the structures of the main products in the following reactions :
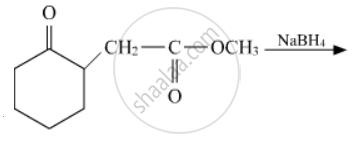
Concept: Importance and Applications of Coordination Compounds
What type of isomerism is exhibited by the complex `[Co(NH_3)_5Cl]SO_4`?
Concept: Isomerism in Coordination Compounds > Structural Isomerism
Write the hybridisation and number of unpaired electrons in the complex `[CoF_6]^(3-)`. (Atomic No. of Co = 27)
Concept: Bonding in Coordination Compounds > Valence Bond Theory (VBT)
Write the chemical reactions involved in the process of extraction of Gold. Explain the role of dilute NaCN and Zn in this process.
Concept: Types of Separation or Concentration of an Ore > Leaching
