Advertisements
Advertisements
प्रश्न
1 J equals to :
पर्याय
0.24 cal
4.18 cal
1 cal
1 kcal
उत्तर
1 J equals to : 0.24 cal
1 calorie = 4.186 J
Therefore,
1 J = 1/4.186 = 0.24 cal
APPEARS IN
संबंधित प्रश्न
What is the Kelvin scale of temperature?
Two bodies at different temperatures are placed in contact. State the direction in which the heat will flow.
Distinguish between heat and temperature.
A body absorbs 1680 J of heat energy. How many calories of heat is absorbed by the body?
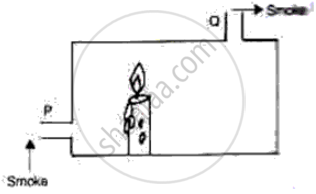
The following figure shows a container in which a lighted candle has been kept. The container has two openings, p, and Q. Which way the smoke from an external source placed near P moves when it reaches P and leaves from Q. Give a reason for your answer.
Explain with a diagram, the effect of the spherical shape of the earth on the temperature at different latitudes.
Why are burns caused by steam more painful than those caused by boiling water at the same temperature?
