Advertisements
Advertisements
प्रश्न
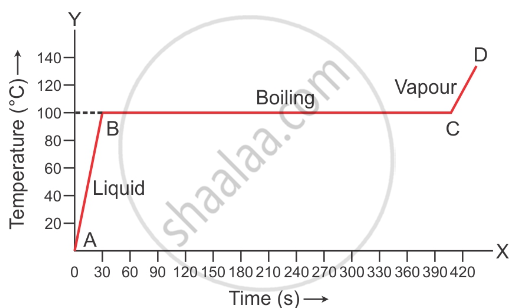
1 kg of ice at 0o is heated at constant rate and its temperature is recorded after every 30 s till steam is formed at 100o C. Draw a temperature time graph to represent the change of phase.
उत्तर
APPEARS IN
संबंधित प्रश्न
What is the principle of Calorimetry?
Differentiate between heat and temperature.
A mass of 200 g of mercury at 100°C is poured into water at 20°C. If the final temperature of the mixture is 25°C, find the mass of water. (Sp. heat cap. of mercury is 140 J/kgK).
Fill in the following blank using suitable word:
Two bodies in contact are said to be in thermal equilibrium, if they have same ...........
Fill in the following blank using suitable word:
Evaporation causes ..........
Which has more heat: 1 g of ice at 0°C or 1 g of water at 0°C? Give reason.
A Bunsen burner raises the temperature of 500g of water from 10°C to 100°C in 5 minutes. What heat is supplied per second?
50 g of ice at 0°C is added to 300 g of a liquid at 30°C. What will be the final temperature of the mixture when all the ice has melted? The specific heat capacity of the liquid is 2.65 Jg-1 °C-1 while that of water is 4.2 J g-1 °C-1. Specific latent heat of fusion of ice = 336 Jg-1.
If you apply equal amount of heat to a solid, liquid, and gas individually, which of the following will have more expansion?
The process of converting a substance from gaseous state to solid state is called _______.
