Advertisements
Advertisements
प्रश्न
88Ra226 experiences three α - decay. Find the number of neutrons in the daughter element.
उत्तर
Given: Mass number of 88Ra226 = 226
Atomic number of 88Ra226 = 88
Mass number of α = 4
Atomic number of α = 2
After 3 α decay, Mass number of the daughter element will be,
= 226 − (3 × 4)
= 226 − 12
= 214
Atomic number of daughter element is,
= 88 − (3 × 2)
= 88 − 6
= 82
∴ Number of neutrons = 214 − 82
= 132
APPEARS IN
संबंधित प्रश्न
Answer the following questions based on a hot cathode ray tube
Name the charged particles
State the energy conversion taking place in a cathode ray tube
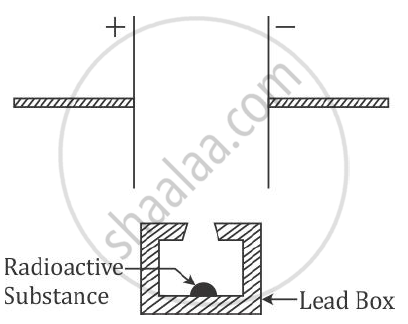
Complete the diagram as given above by drawing the deflection of radioactive radiations in an electric field
State two similarities and two dissimilarities between the γ-rays and X-rays.
What happens to the mass number of an element when (i) an α -particle, (ii) a β -particle, and (iii) γ -radiation is emitted?
Define the term radioactivity. Of the three normally occurring radioactive radiations:
(a) Which has a positive charge?
(b) Which is most penetrating?
(c) Which has no electric charge?
Name a common device where a hot cathode ray tube is used.
Two radioactive nuclei are represented by xAp and yAq and when p and q are mass numbers and x, y are the atomic number. How can the products be represented, i .e., what are the new values of p, q. x and y after the emission of an α-particle and a β-particle from A and B respectively.
A radioactive nucleus \[\ce{^A_ZX}\] undergoes spontaneous decay in the sequence \[\ce{^A_ZX -> _{Z-1 }B -> _{Z-3}C -> _{Z-2}D }\], where Z is the atomic number of element X. The possible decay particles in the sequence are ______.
