Advertisements
Advertisements
प्रश्न
A Carnot engine whose efficiency is 45% takes heat from a source maintained at a temperature of 327°C. To have an engine of efficiency of 60% what must be the intake temperature for the same exhaust (sink) temperature?
उत्तर
Efficiency of Carnot engine (η1) = 45% = 0.45
Initial intake temperature (T1) = 327°C = 600 K
New efficiency (η2) = 60% = 0.6
Efficiency of Carnot engine is given by
η = `1 - "T"_2/"T"_1`
T1 is the temperature of the source; T2 is the temperature of the sink
1st Case: T2 = (1 − η)T1 = (1 − 0.45) × 600
T2 = 330 K
2nd Case: `"T"_2/"T"_1 = 1 - η`
T1 = `"T"_2/(1 - η)`
= `330/(1 - 0.6)`
= `330/0.4`
T1 = 825 K
= 825 − 273
T1 = 552°C
APPEARS IN
संबंधित प्रश्न
A gas contained in a cylinder surrounded by a thick layer of insulating material is quickly compressed has there been a transfer of heat?
Draw a p-V diagram and explain the concept of positive and negative work. Give one example each.
The figure shows the V-T diagram for one cycle of a hypothetical heat engine which uses the ideal gas. Draw the p-V diagram diagram of the system.
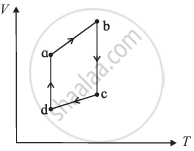
State Kelvin-Planck's statement of the second law of thermodynamics.
Define heat engine.
Explain the heat engine and obtain its efficiency.
Explain in detail the Carnot heat engine.
Heat engine transfers ______.
A heat engine operates between a cold reservoir at temperature T2 = 400 K and a hot reservoir at temperature T1. It takes 300 J of heat from the hot re ervoir and delivers 240 J of heat to the cold reservoir in a cycle. The minimum temperature of the hot reservoir has to be ______ K.
Draw a neat labelled P - V diagram for a typical heat engine.
