Advertisements
Advertisements
प्रश्न
A coordination compound has the formula \[\ce{CoCl3.4NH3}\]. It precipitates silver ions as AgCl and its molar conductance corresponds to a total of two ions.
Based on this information, answer the following question:
- Deduce the structural formula of the complex compound.
- Write the IUPAC name of the complex compound.
- Draw the geometrical isomers of the complex compound.
उत्तर
(a) Because just one AgCl is created, only one chloride ion is precipitated, which equals one negative ion. As a result, the structural formula for the complex will be.
\[\ce{[Co(NH3)4Cl2]Cl}\]
(b) IUPAC name: tetraamminedichloridocobalt (Ill) chloride
(c) Geometrical isomers
\[\begin{array}{cc}
\phantom{..........}\ce{H3N\phantom{....}NH3\phantom{....}Cl\phantom{........}H3N\phantom{....}NH3\phantom{....}Cl\phantom{.........}H3N\phantom{....}Cl\phantom{....}NH3}\phantom{....}\\
\phantom{......}\backslash\phantom{.....}|\phantom{.....}/\phantom{..............}\backslash\phantom{.....}|\phantom{.....}/\phantom{..............}\backslash\phantom{.....}|\phantom{.....}/\phantom{}\\
\phantom{.........}\ce{Co\phantom{........................}Co\phantom{.......................}Co}\phantom{..}\\
\phantom{......}/\phantom{.....}|\phantom{.....}\backslash\phantom{..............}/\phantom{.....}|\phantom{.....}\backslash\phantom{..............}/\phantom{.....}|\phantom{.....}\backslash\phantom{}\\
\phantom{.........}\ce{H3N\phantom{.....}NH3\phantom{....}Cl\phantom{.........}Cl\phantom{.....}NH3\phantom{....}H3N\phantom{........}H3N\phantom{...}NH3\phantom{...}Cl}\phantom{.....}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Account for the following:
Cu+2 salts are coloured, while Zn2+ salts are white.
The elements of 3d transition series are given as: Sc Ti V Cr Mn Fe Co
Answer the following: Which element has the highest m.p?
Explain briefly how +2 state becomes more and more stable in the first half of the first row transition elements with increasing atomic number?
To what extent do the electronic configurations decide the stability of oxidation states in the first series of the transition elements? Illustrate your answer with examples.
What are alloys?
How is potassium dichromate prepared from chrome iron ore?
Metallic radii of some transition elements are given below. Which of these elements will have highest density?
| Element | \[\ce{Fe}\] | \[\ce{Co}\] | \[\ce{Ni}\] | \[\ce{Cu}\] |
| Metallic radii/pm | 126 | 125 | 125 | 128 |
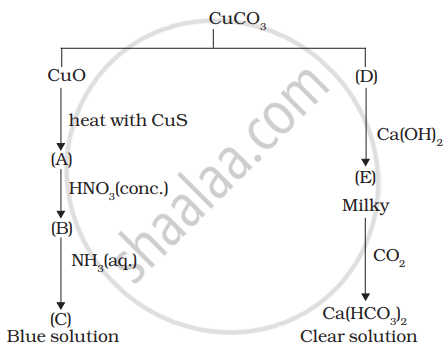
Identify A to E and also explain the reactions involved.
The oxidation state of Fe in [Fe(CO)5] is ______.
Assertion (A): Transition metals have high enthalpy of atomisation.
Reason (R): Greater number of unpaired electrons in transition metals results in weak metallic bonding.
