Advertisements
Advertisements
प्रश्न
A reddish-brown metal ‘X’, when heated in air, gives a black compound ‘Y’, which when heated in the presence of H2 gas gives ‘X’ back. ‘X’ is refined by the process of electrolysis; this refined form of ‘X’ is used in electrical wiring. Identify ‘X’ and ‘Y’. Draw a well-labelled diagram to represent the process of refining ‘X’.
उत्तर
- ‘X’ - Copper/Cu and ‘Y’ - CuO.
- The diagram represents the process of refining ‘X'.
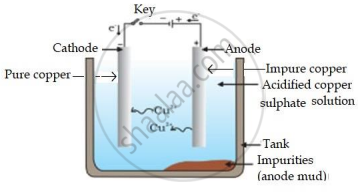
APPEARS IN
संबंधित प्रश्न
How are metals refined by the electrolytic process? Describe the electrolytic refining of copper with the help of a neat labelled diagram.
Draw a neat labelled diagram.
Magnetic separation method.
Which of the following process to be carried out to avoid the
formation of greenish layer on brass vessels due to corrosion?
Electrolysis is used to obtain pure metal from impure metal.
The lightest liquid metal is ____________.
Bauxite is mixed with cryolite so as to
(i) reduce its melting point
(ii) increase its electrical conductivity
(iii) molten cryolite acts as a solvent
(iv) increase its melting point
Metals are refined by using different methods. Which of the following metals are refined by electrolytic refining?
(i) Au
(ii) Cu
(iii) Na
(iv) K
Reaction between X and Y, forms compound Z. X loses electron and Y gains electron. Which of the following properties is not shown by Z?
Give the formulae of the stable binary compounds that would be formed by the combination of following pairs of elements.
- Mg and N2
- Li and O2
- Al and Cl2
- K and O2
Answer the following question with reference to the electrorefining of copper:
What do you observe at the cathode?
