Advertisements
Advertisements
प्रश्न
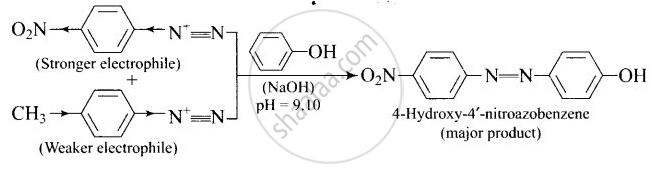
A solution contains 1 g mol. each of p-toluene diazonium chloride and p-nitrophenyl diazonium chloride. To this 1 g mol. of alkaline solution of phenol is added. Predict the major product. Explain your answer.
उत्तर
This reaction is an example of electrophilic aromatic substitution. In alkaline medium, phenol forms phenoxide ion which is more electron-rich than phenol and hence more reactive for electrophilic attack. The electrophile in this reaction is aryl diazonium cation. p-Nitrophenyldiazonium cation is a stronger electrophile than p-toluene diazonium cation. Therefore, it couples preferentially with phenol.
APPEARS IN
संबंधित प्रश्न
What is the action of the following reagents on aniline?
Bromine water
Give reasons for the following:
Aniline does not undergo Friedel- Crafts reaction.
Write the structures of main products when aniline reacts with the following reagents :
Br2 water
Write short notes on acetylation.
What is the action of acetic anhydride on diethylamine?
What is the action of the following reagents on aniline?
Acetic anhydride
Assertion: N, N-Diethylbenzene sulphonamide is insoluble in alkali.
Reason: Sulphonyl group attached to nitrogen atom is strong electron-withdrawing group.
When bromination of aniline is carried out by protecting – NH2. The major product is
In the following reaction, the reason for why meta-nitro product also formed is:

Aniline does not give Friedel-Crafts reaction. Give a reason.
