Advertisements
Advertisements
प्रश्न
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution:
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | _________ | _________ |
| 2. | Magnesium ribbon | _________ | _________ |
| 3. | Manganese (IV) oxide with heating | _________ | _________ |
| 4. | Sodium sulphide | _________ | _________ |
Complete the table by writing the gas evolved in each case and its odour.
उत्तर
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | \[\ce{CO2}\] | Odourless |
| 2. | Magnesium ribbon | \[\ce{H2}\] | Odourless |
| 3. | Manganese (IV) oxide with heating | \[\ce{Cl2}\] | Strong Pungent odour |
| 4. | Sodium sulphide | \[\ce{H2S}\] | Rotten egg |
APPEARS IN
संबंधित प्रश्न
For the preparation of hydrochloric acid in the laboratory:
What arrangement is done to dissolve hydrogen chloride gas in water?
Give three distinct test [apart from using an indicator] you would carry out with solution of \[\ce{HCl}\] to illustrate the typical properties of an acid.
State which component is the oxidizing agent in aqua regia.
Name the following:
Five oxidizing agents that liberated chlorine from concentrated hydrochloric acid.
What is the function of HCI in preparation of aqua-regia?
Write a fully balanced of the following case :
Red lead is warmed with concentrated hydrochloric acid.
How will you distinguish between the two black samples, CuO and MnO2 with a chemical test?
Match the following:
| Column A | Column B |
| 1. A substance that turns moist starch iodide paper blue. | A. Ammonium sulphate |
| 2. A compound which release a reddish brown gas on reaction with concentrated sulphuric acid and copper turnings. | B. Lead carbonate |
| 3. A solution of this compound gives dirty green precipitate with sodium hydroxide. | C. Chlorine |
| 4. A compound which on heating with sodium hydroxide produces a gas which forms dense white fumes with hydrogen chloride. | D. Copper nitrate |
| 5. A white solid which gives a yellow residue on heating | E. Ferrous sulphate |
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
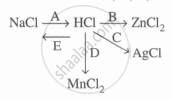
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.