Advertisements
Advertisements
प्रश्न
Solve the following question.
A steady current of 2 amperes was passed through two electrolytic cells X and Y connected in series containing electrolytes FeSO4 and ZnSO4 until 2.8 g of Fe deposited at the cathode of cell X. How long did the current flow? Calculate the mass of Zn deposited at the cathode of cell Y.
(Molar mass : Fe = 56 g mol–1, Zn = 65.3 g mol–1, 1F = 96500 C mol–1)
उत्तर
Given: I = 2A
(i)
Fe2+ 2e– → Fe
56 g of Fe requires = 2 × 96500 C charge
2.8 g of Fe requires = `(2 xx 96500)/(56)` x 2.8 C charge
Q = 9650
Q = It
`"t" = "Q"/"I" = (9650)/(2)` = 4825 sec
(ii)
Zn2+ + 2e– → Zn
2 x 96500 C of electricity deposit Zn = 65.3 g
1 C of elecrticity deposit Zn = `(65.3)/(2 xx 96500)`
9650 C of elecrticity deposit Zn = `(65.3)/(2 xx 96500) xx 9650`
= 3.265 g
APPEARS IN
संबंधित प्रश्न
The charge of how many coulomb is required to deposit 1.0 g of sodium metal (molar mass 23.0 g mol-1) from sodium ions is -
- 2098
- 96500
- 193000
- 4196
Write any four applications of electrochemical series
How much charge is required for the following reduction:
1 mol of \[\ce{Cu^{2+}}\] to \[\ce{Cu}\]?
How much charge is required for the following reduction:
1 mol of \[\ce{MnO^-_4}\] to \[\ce{Mn^{2+}}\]?
Calculate the mass of Ag deposited at cathode when a current of 2 amperes was passed through a solution of AgNO3 for 15 minutes.
(Given : Molar mass of Ag = 108 g mol−1 lF = 96500 C mol−1)
According to Faraday’s First Law of Electrolysis, the amount of chemical reaction which occurs at any electrode during electrolysis by a current is proportional to the ____________.
Consider the figure and answer the following question.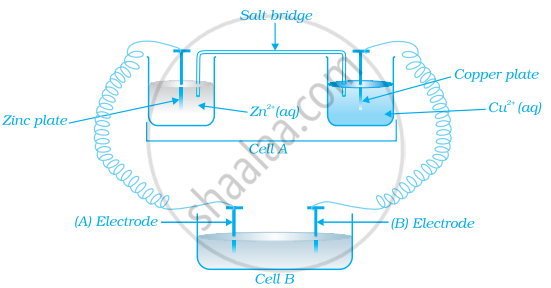
Cell ‘A’ has ECell = 2V and Cell ‘B’ has ECell = 1.1V which of the two cells ‘A’ or ‘B’ will act as an electrolytic cell. Which electrode reactions will occur in this cell?
Given `1/a` = 0.5 CM–1, R = 50 ohm, N = 1.0 then equivalent conductance of electrolytic cell is
On Electrolysis of dilute sulphuric acid using platinum electrodes, the product obtained at the anode will be.
What is the quantity of electricity in Coulombs required to produce 4.8 g of Mg from molten MgCl2? How much Ca will be produced if the same amount of electricity was passed through molten CaCl2? (Atomic mass of Mg = 24 u, atomic mass of Ca = 40 u).
