Advertisements
Advertisements
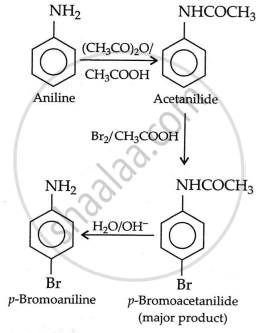
प्रश्न
Accomplish the following conversion:
Aniline to p-bromoaniline
उत्तर
APPEARS IN
संबंधित प्रश्न
Arrange the following in increasing order of their basic strength :
C6H5 – NH2, C6H5 – CH2 – NH2, C6H5 – NH – CH3
- Write structures of different isomeric amines corresponding to the molecular formula C4H11N.
- Write the IUPAC names of all the isomers.
- What type of isomerism is exhibited by different pairs of amines?
How will you convert Benzene into N, N-dimethylaniline?
Arrange the following in increasing order of their basic strength:
C2H5NH2, C6H5NH2, NH3, C6H5CH2NH2 and (C2H5)2NH
Account for the following:
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Arrange the following:
In increasing order of boiling point:
C2H5OH, (CH3)2NH, C2H5NH2
Write a short note on ammonolysis.
Give plausible explanation for each of the following:
Why are aliphatic amines stronger bases than aromatic amines?
Complete the following reaction:
\[\ce{C6H5N2Cl + H3PO2 + H2O ->}\]
The CORRECT decreasing order of solubility in water will be ____________.
Which among the following has the highest boiling point?
Among the following isomeric amines, an amine having highest boiling point is:
Assertion: N-Ethylbenzene sulphonamide is soluble in alkali.
Reason: Hydrogen attached to nitrogen in sulphonamide is strongly acidic.
Acetic acid exist as dimer in benzene due to
Dimerisation of carboxylic acids is due to
The hydrogen bond is shortest in
Which of the following amines form maximum hydrogen bonds within themselves?
Arrange the following in increasing order of their boiling point:
C2H5OH, C2H5NH2, (C2H5)3N
Arrange the decreasing boiling point.
\[\ce{CH3COOH, C2H5OH, CH3NH2, CH3OCH3}\]
